Indonesia
Regulatory Consultant Office Profile
Regulatory Consultant Office Profile
Our office in Jakarta, Indonesia, is staffed by diligent and highly experienced regulatory professionals whose primary goal is to get you market access all whilst remaining compliant.
With the 4th largest population in the world and a government commitment to invest more in healthcare, Indonesia is one of the most ideal nations to distribute Medical Devices. The Indonesia Ministry of Health has a streamlined Medical Device Registration and post-market surveillance through their eCatalogue system. Given the geographical diversity of the country (it sits in 4 time zones!), having Asia Actual as your Independent License Holder (ILH) enables more widespread distribution of your Medical Device or IVD. With an ILH, manufacturers can use multiple distributors to expand their reach, rather than just being locked in with one.

Ilham has been employed with Asia Actual Indonesia since July, 2018. He has over 10 years of experience managing device registrations and quality systems in Indonesia for multinational companies such as Drager Medical (Germany), Nipro Medical (Japan), and Bu Kwang Medical (Korea). His portfolio of diverse registration work includes electromedical (radiation and non-radiation emitting) and non-electromedical (sterile and non-sterile) devices as well as IVD reagents.
Ilham holds a Bachelor’s degree from the Bandung School of Pharmacy and a Master’s degree in Management from Widyatama University (Badung). Additionally, Ilham is active in advocacy and engagement initiatives through industry bodies such as the Medical Device and Laboratory Equipment Association (GAKESLAB). He speaks Bahasa Indonesian and English.

Ridwan cofounded Indonesia with Bryan in 2016 and has been in the Medical Device Industry since 1981. Ridwan enjoys the challenges of the field. Given the rate at which Medical Technology has grown and become more advanced, so too must regulations and trading systems to accommodate such progress. Because of this, he is adamant about staying up-to-date on the latest developments in Med-Tech as well as in Indonesian and ASEAN regulations.
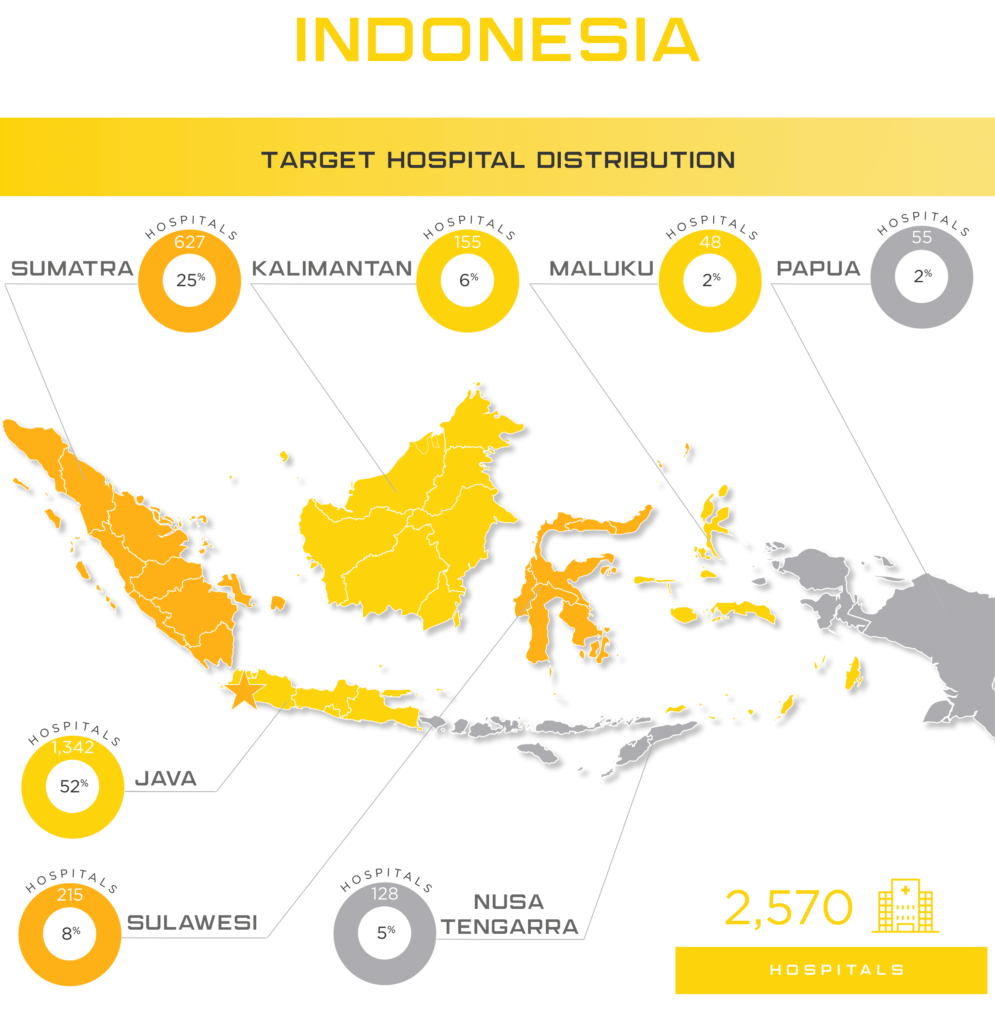
Asia Actual is available to help navigate the complex medical device registration requirements and regulatory pathway for medical device and IVD distribution in Indonesia. Contact Asia Actual for a free consultation discussing the potential for your medical device or IVD in the Indonesia medical device market.

US: +1 512 898-9222
SG: +65 8800-3197
EMAIL: Indonesia@asiaactual.com
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
116 Changi Road, #04-05
