Contact Us
FREE Consultation.
PHONE: 512-898-9222
EMAIL: inquiry@asiaactual.com
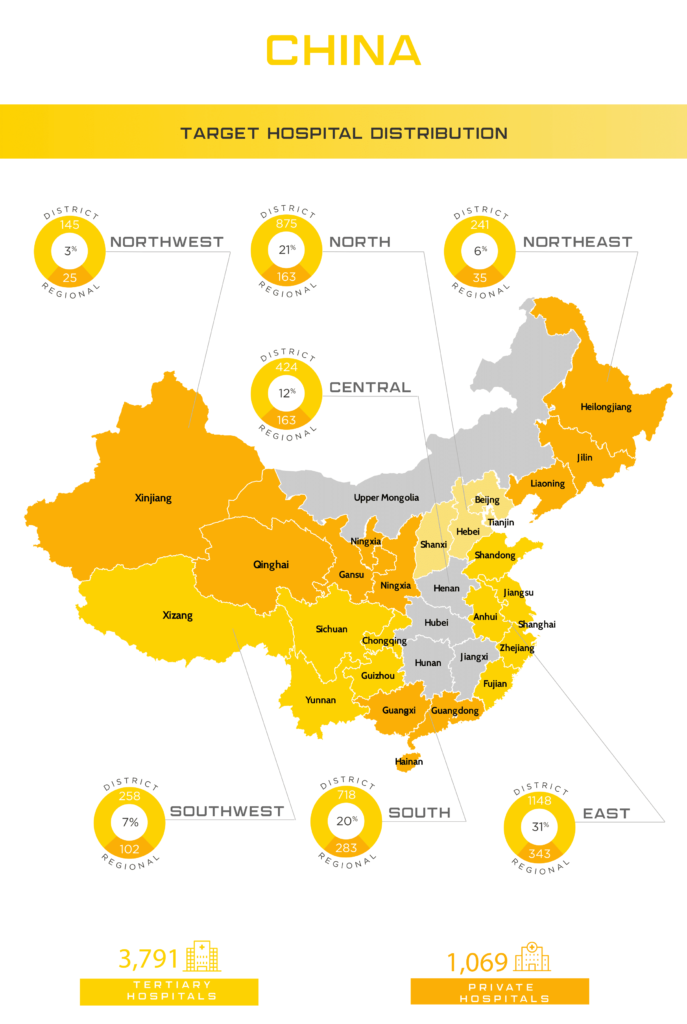
Currently with the largest population in the world, China remains a delicate market to break into for foreign medical device manufacturers. As China incrementally opens their lucrative market to foreign companies, these manufacturers still face complex hurdles when it comes to identifying effective partnerships for distributing their products locally, while maintaining sufficient protections to their intellectual property. This being said, these hurdles are far easier to overcome today and will likely continue to trend in this direction as China becomes more developed and seeks to have their own local companies gain equal access and IP protections beyond their home market.
As China’s population has rapidly been pulled from below the poverty line the past few decades, the government is now shifting some of their efforts from focusing solely on basic necessities to finding ways to continuously improve the lives of their people. As such, by 2018, China’s healthcare expenditures grew to 5.7% of their GDP, translating into a $777 billion industry. On top of this, the medical device market is only 14% of their pharmaceuticals market, compared to 42% of the global market, showing an immense opportunity for medical device manufacturers as China’s healthcare market continues to evolve towards 1st world status.
Asia Actual is available to help navigate the complex medical device registration requirements and regulatory pathway for medical device and IVD distribution in China.
Contact Asia Actual for a free consultation discussing the potential for your medical device or IVD in the Chinese market.

FREE Consultation.
PHONE: 512-898-9222
EMAIL: inquiry@asiaactual.com
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
116 Changi Road, #04-05
