Contact Us
US: +1 512 898-9222
SG: +65 8800-3197
EMAIL: Inquiry@asiaactual.com
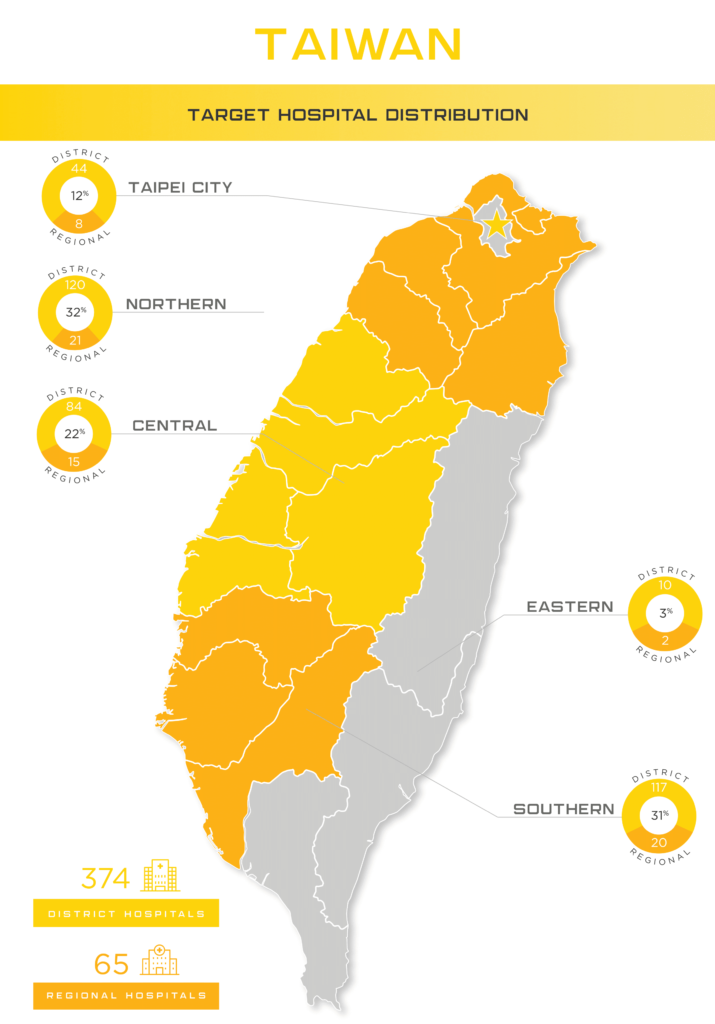
While Taiwan has a small island population of 23 million, its medical device market potential should not be overlooked. Given the limited domestic manufacturing capabilities, Taiwan has a high demand for importing advanced medical devices. Over 70 percent of the market is supplied by imports, primarily coming from the United States. Also, given Taiwan’s location, it is a hub of transportation with easy access to surrounding countries. This is beneficial for manufacturers who may also be interested in entering larger markets, such as mainland China.
Additionally, around 15% of the Taiwanese population is either age 65 or older, indicating an aging population with more terminal diseases. Therefore, there is high demand for products such as artificial joints and cardiovascular equipment. Taiwan’s government is also increasing healthcare expenditure to address this growing concern, spending $41.3 billion in 2019.
The Taiwanese medical device market can be complex, but Asia Actual has experienced, local, bi-lingual consultants to answer any questions about the regulatory process.
Asia Actual is available to help navigate the complex medical device registration requirements and regulatory pathway for medical device and IVD distribution in Taiwan
Contact Asia Actual for a free consultation discussing the potential for your medical device or IVD in the Taiwanese market.

US: +1 512 898-9222
SG: +65 8800-3197
EMAIL: Inquiry@asiaactual.com
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
116 Changi Road, #04-05
