Thailand Medical Device Market
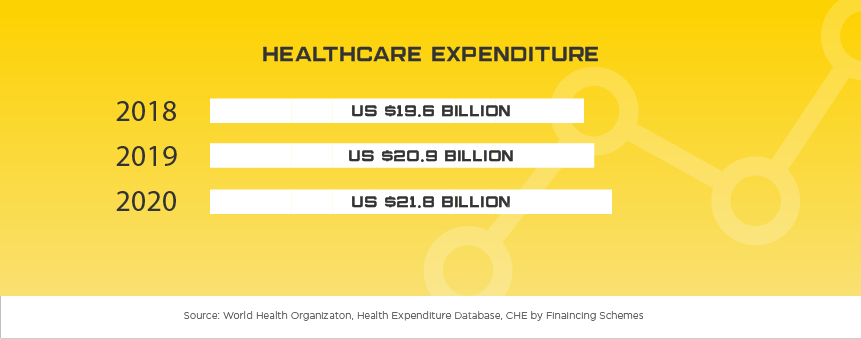
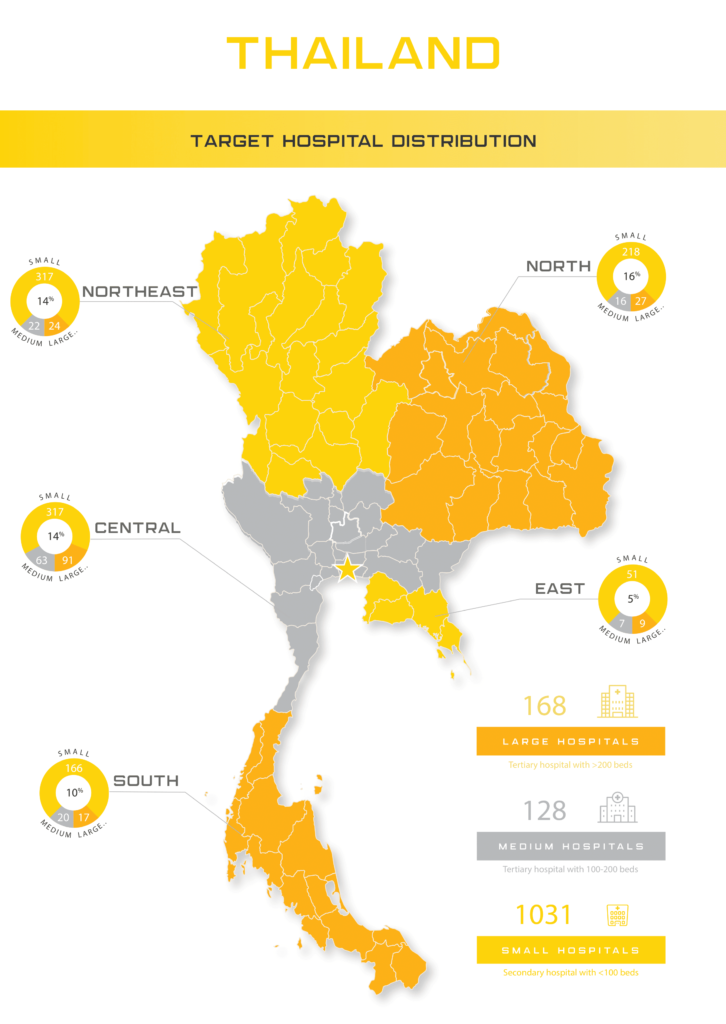
Estimated at US$2.1 billion in 2018, Thailand heavily relies on foreign imports for the vast majority of their medical device needs. Compared to other countries in the region, Thailand can be seen as successfully developing their own medical device manufacturing sector by targeting primarily consumable products where they can better compete in the local and neighboring markets.
While Singapore may still have the top medical tourism industry in southeast Asia, Thailand is currently seeing a surge in this industry. Along with providing a cost effective alternative to the large populations of many nearby countries, the low language barrier and “Westernization” of their medical centers indicates Thailand should be able to sustain their recent growth. Additionally, to support the continued growth in this sector, the Thai Board of Investment (BOI) provides local startups and foreign investors with various incentives to spur ongoing investment and development.
Grow With Us
Asia Actual is available to help navigate the complex medical device registration requirements and regulatory pathway for medical device and IVD distribution in Thailand.
Contact Asia Actual for a free consultation discussing the potential for your medical device or IVD in the Thailand medical device market.
Important Documents and Links
Thailand Regulatory Support
Local Office

Latest Market Updates
 Software as a Medical Device in ThailandJune 17, 2024 - 1:17 pm
Software as a Medical Device in ThailandJune 17, 2024 - 1:17 pm Thai FDA Regulatory Documentation ExemptionsApril 12, 2024 - 4:00 pm
Thai FDA Regulatory Documentation ExemptionsApril 12, 2024 - 4:00 pm Thai FDA Reduces Application Review TimeMarch 13, 2024 - 11:29 am
Thai FDA Reduces Application Review TimeMarch 13, 2024 - 11:29 am Thai FDA QMS Updates to Affect GDP, GMP, and GCPFebruary 14, 2024 - 9:36 pm
Thai FDA QMS Updates to Affect GDP, GMP, and GCPFebruary 14, 2024 - 9:36 pm Thailand FDA to Decrease Registration Application Processing TimesJanuary 25, 2024 - 12:34 pm
Thailand FDA to Decrease Registration Application Processing TimesJanuary 25, 2024 - 12:34 pm
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
Asia Actual, LLC
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
Asia Headquarters
116 Changi Road, #04-05