Contact Us
US: +1 512 898-9222
SG: +65 3138-4148
EMAIL: Thailand@asiaactual.com
Importing medical devices into Thailand is more difficult than in other countries in the region. It is important to work with an importer with staff experienced with medical device imports to avoid customs clearance delays and added costs.
Unique to the customs clearance process for medical devices in Thailand is the requirement for a License Per Invoice (LPI) approved by the Thai FDA for each shipment. The Import License holder must apply with credentials, the original Import License, and original shipping documents (e.g., Air Waybill, shipping invoice and packing list) submitted online to Thai FDA. (The one-time fee to the TFDA for LPI submission which includes the Stamp Duty, Power of Attorney, and document submission). The FDA will confirm that all devices included in the shipment (by part number) are covered by a valid import license and then issue the LPI. The application review process take 1 to 2 days. The resulting License Per Invoice document is a required element of the customs clearance process for registered medical devices, accessories and spare parts.
The overall process is then similar as in other countries with assemble a submission of shipping documents, payment of import tax (if any depending on HS code), payment of VAT (7% on all goods and services), etc. Shipping rate will vary significantly by mode and point of origin; and should be known to manufacturers.
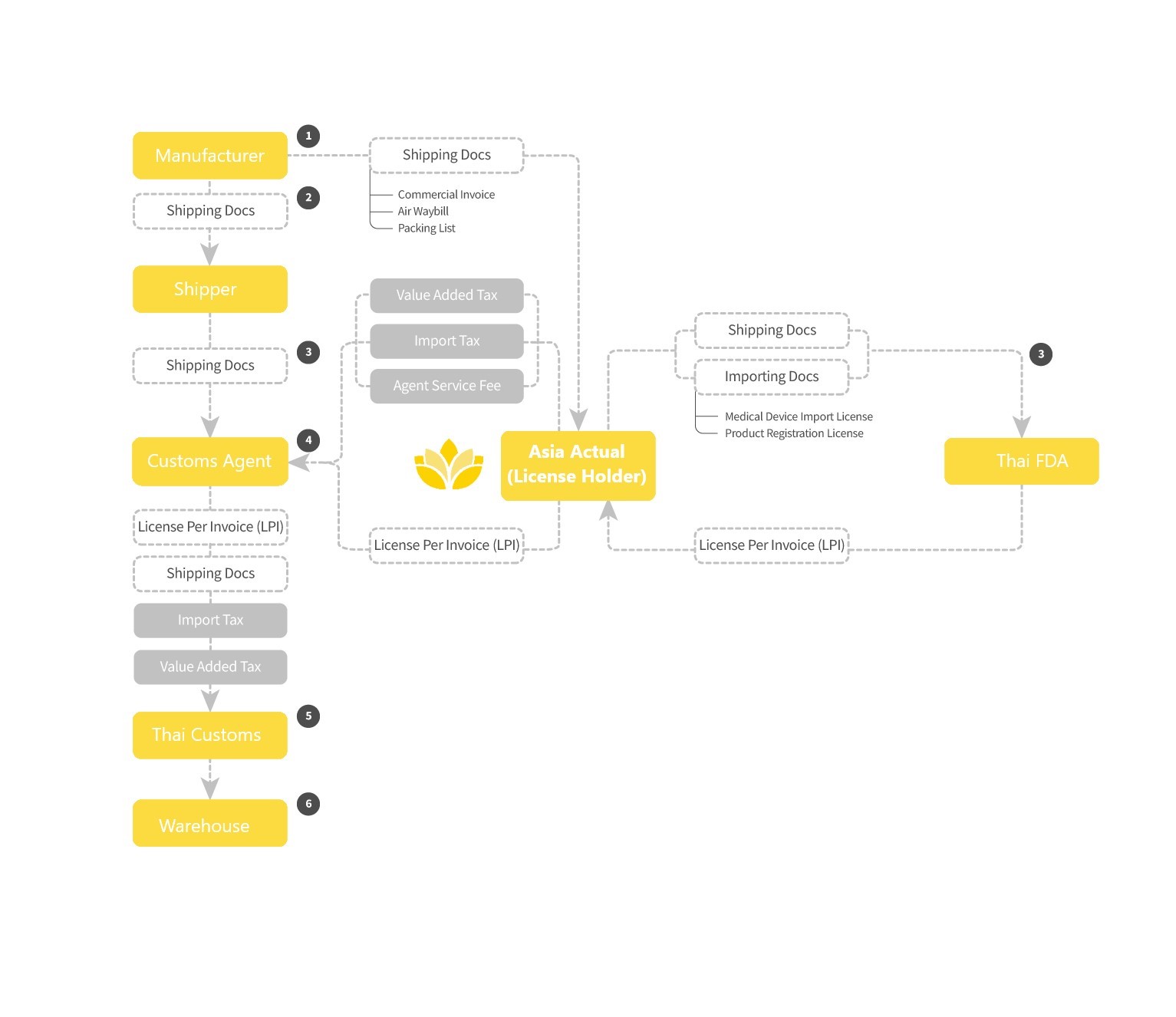
A source of common delays can be:
In Thailand, the License Holder must be involved in the customs clearance process for devices for which it holds the Import License. Asia Actual is committed to fair, efficient and transparent importing transactions. Competitively bid third-party services (e.g., freight forwarding) are charged on a pass-through basis. A Shipment Report is generated per event to record fees and document receipts.
Basic support from Asia Actual will involve taking title of the shipment through customs clearance and managing the following activities:
Optional support services are available such as long-term storage, break-bulk, labeling, and order fulfillment.
Using Asia Actual as your License Holder in Thailand comes with the added benefit of expertly managed, efficient and transparent importations.
Contact Asia Actual for more information on our importing services.
US: +1 512 898-9222
SG: +65 3138-4148
EMAIL: Thailand@asiaactual.com
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
116 Changi Road, #04-05
