Contact Us
US: +1 512 898-9222
SG: +65 3138-4148
EMAIL: Hongkong@asiaactual.com
Medical device labeling requirements in Hong Kong for listed devices are laid out in the Special Listing Labeling. In addition to information on the international label, Hong Kong requires the HK listing number and the contact details of the Local Responsible Person (LRP) in English and Chinese (Kaishu font type with and all characters should not be less than 2mm high) text.
See 4.4.13 Special Listing Information in the GN-01
The Special Listing Information of a medical device comprises (i) and (ii) below:
The LRP shall provide the Special listing Information by complying with either Option (I) or Option (II) below. The LRP will have a grace period of six months after the device is listed to meet this requirement.
Option (I)
Option (II)
For additional Medical Device labeling requirements please visit the MDACS (Medical Device Administrative Control System) Technical Reference document (TR-005:2021(E)).
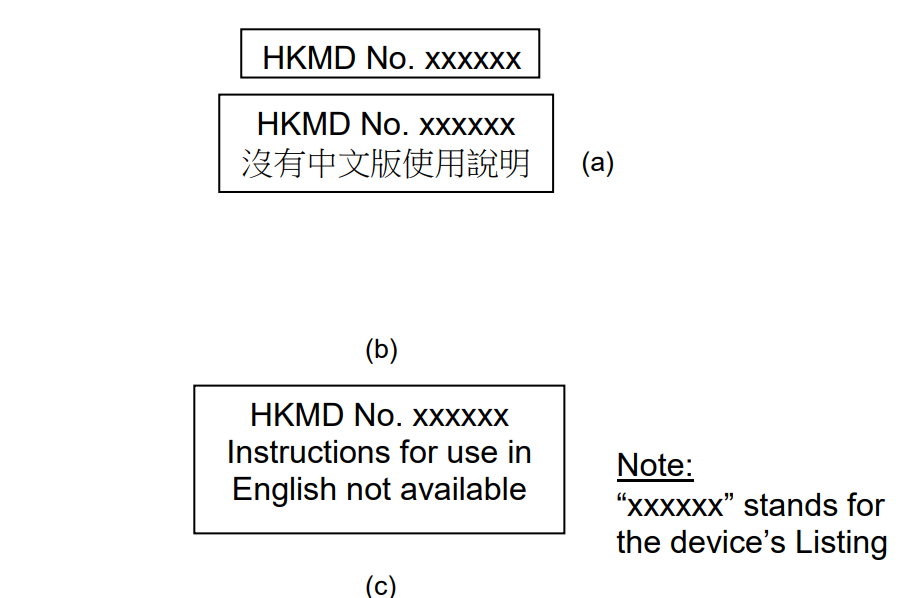
Fig 1. If the instructions for use are available in both English and Chinese languages, the format in figure (a) shall be applied. The format in figure (b) or (c) shall be applied if the instructions for use are available only in Chinese or only in English.
Whenever figure (a), (B) or (c) is applied, it shall be with a printed rectangular border as shown. All the characters shall be of a uniform font size of not less than 2mm high. In (c) the Chinese characters shall be in kaishu.
A family is a group of medical devices having the same manufacturer, device description and classification, intended use, design, construction and performance e.g. catheters of different diameters and lengths. For each member of the medical device family, its identifier(s) (e.g. product number), and a brief account of its characteristics that distinguish it from other members (e.g. dimensions of its various parts) are to be provided.
A system is a medical device comprising a number of medical devices (component medical devices) intended to be used together to fulfil the system’s intended use. All component medical devices shall be placed on the market under the name of the same manufacturer. A short description on how the component medical devices are used together to achieve the intended purpose of the medical device system shall be provided. For each component medical device of a medical device system, please provide its Asian Medical Device Nomenclature System (AMDNS) term (if an AMDNS term is not available for a particular component, a short description shall be provided) and the corresponding AMDNS code, its identifier(s) (e.g. model number), and a brief description of its intended use.
A series is a group of medical devices belonging to the same model series of a manufacturer and having the same device classification and intended use, but differing only in minor features or functions that do not present significantly different safety, performance, and effectiveness issues. In principle, the designs, labeling, and manufacturing processes and performance specifications cannot be significantly different between members of a series. For each member of the medical device series, its identifier(s) (e.g. product number), and a brief account of its characteristics that distinguish it from other members (e.g. dimensions of its various parts) are to be provided.
Additionally, three general rules of grouping apply to each category:
Any changes/updates to the Instructions for Use (IFU) or the device labeling will require an amendment application to be submitted to the MDD.
Contact Asia Actual to see if our Hong Kong labeling solutions are right for your needs.
US: +1 512 898-9222
SG: +65 3138-4148
EMAIL: Hongkong@asiaactual.com
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
116 Changi Road, #04-05
