Singapore Medical Device Market
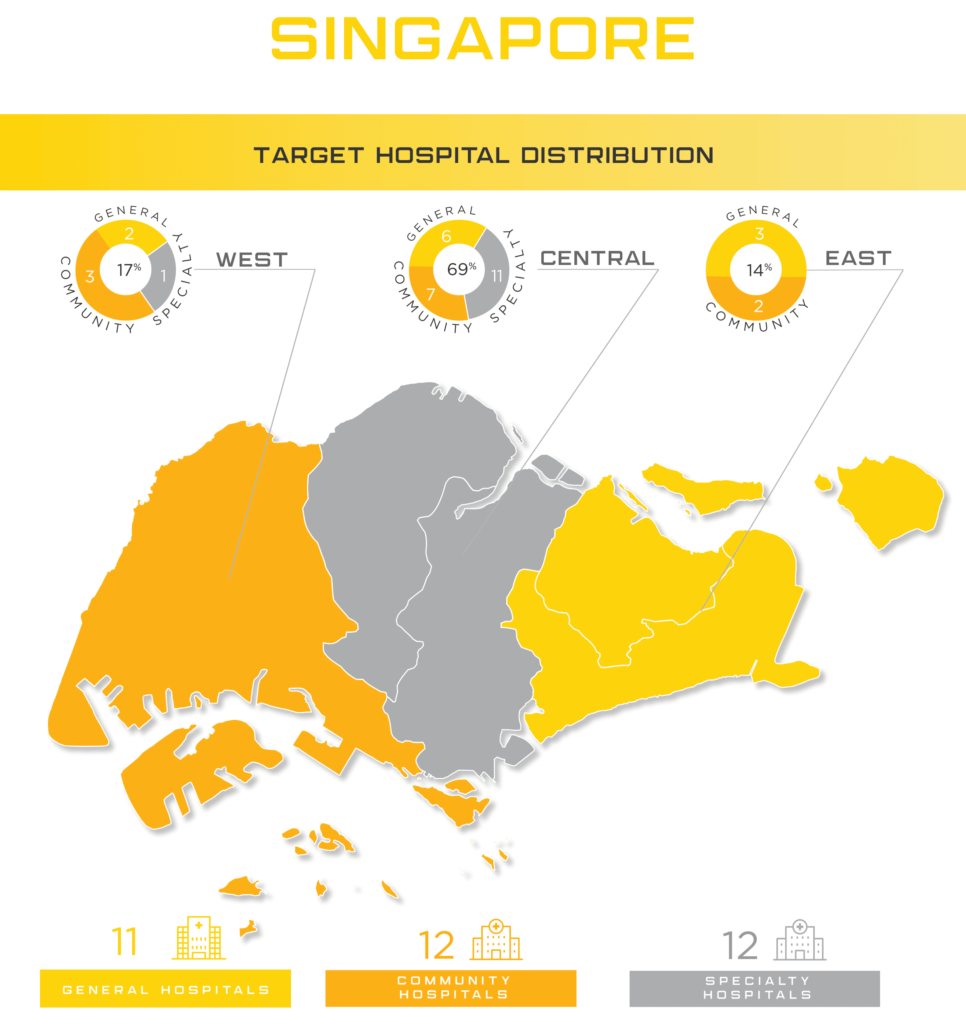
Singapore is renowned for its role as a healthcare hub for the region and is overseen by the Health Science Authority (link). With patients traveling from neighboring countries, Singapore draws over 400,000 patients to receive its high-quality healthcare. Regularly listed in the top ranks for global healthcare, Singapore continues to be a lucrative medical device market with high demand for healthcare and research communities.
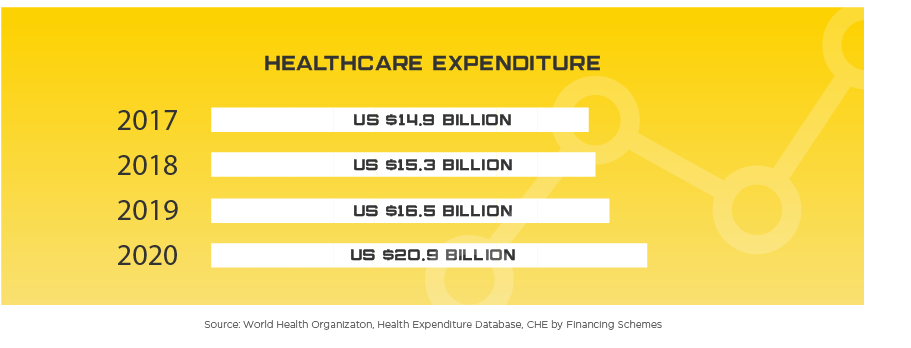
The government is very committed to Singapore’s healthcare needs, installing a long-term plan to raise GDP spending on healthcare to 8%, up from 4.6% currently. The National Health Expenditure is expected to rise to US$9.6 billion per year by 2020. Singapore has strong fundamentals in healthcare excellence, providing strong infrastructure and universal health coverage, further increasing the demand in the Singapore medical device market.
GROW WITH US
Asia Actual is available to help navigate the complex medical device registration requirements and regulatory pathway for medical device and IVD distribution in Singapore.
Contact Asia Actual for a free consultation discussing the potential for your medical device or IVD in the Singapore medical device market.
Important Documents and Links
Singapore Regulatory Support

Kenna Rasiklal
Contact Us
US: +1 512 898-9222
SG: +65 8800-3197
EMAIL: Singapore@asiaactual.com
Latest Market Updates
 How New EU MDR Requirements Will Affect Registrations in AsiaAugust 12, 2022 - 2:07 pm
How New EU MDR Requirements Will Affect Registrations in AsiaAugust 12, 2022 - 2:07 pm Medical Device Advertising Requirements in AsiaJuly 22, 2022 - 10:08 pm
Medical Device Advertising Requirements in AsiaJuly 22, 2022 - 10:08 pm What is CSDT Documentation?July 13, 2022 - 10:27 am
What is CSDT Documentation?July 13, 2022 - 10:27 am Singapore’s HSA Issues New Software as Medical Device (SAMD) GuidanceJuly 6, 2022 - 2:32 pm
Singapore’s HSA Issues New Software as Medical Device (SAMD) GuidanceJuly 6, 2022 - 2:32 pm Singapore’s HSA Removes Classification and Grouping Inquiry OptionApril 1, 2022 - 2:49 pm
Singapore’s HSA Removes Classification and Grouping Inquiry OptionApril 1, 2022 - 2:49 pm
実際の亞洲
เอเชีย แอคชวล
एशिया वास्तविक
실제 아시아
Asia Actual, LLC
515 Congress Avenue, Suite 2100
Austin, TX 78701
+1 512 898-9222
Contact Us
Privacy Policy
Asia Headquarters
116 Changi Road, #04-05